Research Highlights
Revisiting the recombinant history of HIV-1 group M with dynamic network community detection - Poon Lab
 Significance
Significance
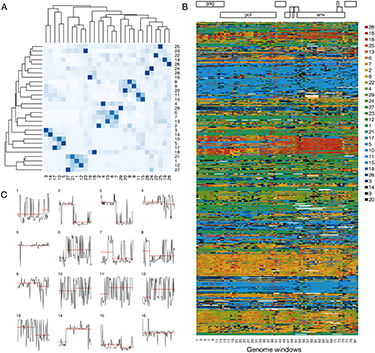
Recombination is a major mechanism through which HIV type 1 (HIV-1) maintains genetic diversity and interferes with viral eradication efforts. There is growing evidence demonstrating a recombinant origin of primate lentiviruses including HIV-1 group M (HIV-1/M). Inferring the extent of recombination across the entire HIV-1/M genome is of great importance as it provides deeper insights into the origin, dynamics, and evolution of the global pandemic. Here we propose an alternative method that can reconstruct the extent of genome-wide recombination in HIV-1, uncover reticulate patterns, and serve as a framework for HIV-1 classification. Our method provides an alternative approach for understanding the roles of virus recombination in the early evolutionary history of zoonosis for other emerging viruses.
Chronic stress physically spares but functionally impairs innate-like invariant T cells - Haeryfar Lab
 The deleterious effects of psychological stress on mainstream T lymphocytes are well documented. However, how stress impacts innate-like T cells is unclear. We report that long-term stress surprisingly abrogates both T helper 1 (TH1)- and TH2-type responses orchestrated by invariant natural killer T (iNKT) cells. This is not due to iNKT cell death because these cells are unusually refractory to stress-inflicted apoptosis. Activated iNKT cells in stressed mice exhibit a “split” inflammatory signature and trigger sudden serum interleukin-10 (IL-10), IL-23, and IL-27 spikes. iNKT cell dysregulation is mediated by cell-autonomous glucocorticoid receptor signaling and corrected upon habituation to predictable stressors. Importantly, under stress, iNKT cells fail to potentiate cytotoxicity against lymphoma or to reduce the burden of metastatic melanoma. Finally, stress physically spares mouse mucosa-associated invariant T (MAIT) cells but hinders their TH1-/TH2-type responses. The above findings are corroborated in human peripheral blood and hepatic iNKT/MAIT cell cultures. Our work uncovers a mechanism of stress-induced immunosuppression.
The deleterious effects of psychological stress on mainstream T lymphocytes are well documented. However, how stress impacts innate-like T cells is unclear. We report that long-term stress surprisingly abrogates both T helper 1 (TH1)- and TH2-type responses orchestrated by invariant natural killer T (iNKT) cells. This is not due to iNKT cell death because these cells are unusually refractory to stress-inflicted apoptosis. Activated iNKT cells in stressed mice exhibit a “split” inflammatory signature and trigger sudden serum interleukin-10 (IL-10), IL-23, and IL-27 spikes. iNKT cell dysregulation is mediated by cell-autonomous glucocorticoid receptor signaling and corrected upon habituation to predictable stressors. Importantly, under stress, iNKT cells fail to potentiate cytotoxicity against lymphoma or to reduce the burden of metastatic melanoma. Finally, stress physically spares mouse mucosa-associated invariant T (MAIT) cells but hinders their TH1-/TH2-type responses. The above findings are corroborated in human peripheral blood and hepatic iNKT/MAIT cell cultures. Our work uncovers a mechanism of stress-induced immunosuppression.
https://news.westernu.ca/2021/04/chronic-stress-cancer/
Coagulase-negative staphylococci release a purine analog that inhibits Staphylococcus aureus virulence - Heinrichs Lab

Coagulase-negative staphylococci and Staphylococcus aureus colonize similar niches in mammals and conceivably compete for space and nutrients. Here, we report that a coagulase-negative staphylococcus, Staphylococcus chromogenes ATCC43764, synthesizes and secretes 6-thioguanine (6-TG), a purine analog that suppresses S. aureus growth by inhibiting de novo purine biosynthesis. We identify a 6-TG biosynthetic gene cluster in S. chromogenes and other coagulase-negative staphylococci including S. epidermidis, S. pseudintermedius and S. capitis. Recombinant S. aureus strains harbouring this operon produce 6-TG and, when used in subcutaneous co-infections in mice with virulent S. aureus USA300, protect the host from necrotic lesion formation. Used prophylactically, 6-TG reduces necrotic skin lesions in mice infected with USA300, and this effect is mediated by abrogation of toxin production. RNAseq analyses reveal that 6-TG downregulates expression of genes coding for purine biosynthesis, the accessory gene regulator (agr) and ribosomal proteins in S. aureus, providing an explanation for its effect on toxin production.
https://news.westernu.ca/2021/04/new-tool-fights-antibiotic-infections/
"Understanding antibodies at play in long-lasting COVID-19 immunity." - Dikeakos and Dekaban Labs
 Characterization of the humoral response to SARS-CoV-2, the etiological agent of COVID-19, is essential to help control the infection. The neutralization activity of plasma from patients with COVID-19 decreases rapidly during the first weeks after recovery. However, the specific role of each immunoglobulin isotype in the overall neutralizing capacity is still not well understood. In this study, we select plasma from a cohort of convalescent patients with COVID-19 and selectively deplete immunoglobulin A, M, or G before testing the remaining neutralizing capacity of the depleted plasma. We find that depletion of immunoglobulin M is associated with the most substantial loss of virus neutralization, followed by immunoglobulin G. This observation may help design efficient antibody-based COVID-19 therapies and may also explain the increased susceptibility to SARS-CoV-2 of autoimmune patients receiving therapies that impair the production of immunoglobulin M (IgM).
Characterization of the humoral response to SARS-CoV-2, the etiological agent of COVID-19, is essential to help control the infection. The neutralization activity of plasma from patients with COVID-19 decreases rapidly during the first weeks after recovery. However, the specific role of each immunoglobulin isotype in the overall neutralizing capacity is still not well understood. In this study, we select plasma from a cohort of convalescent patients with COVID-19 and selectively deplete immunoglobulin A, M, or G before testing the remaining neutralizing capacity of the depleted plasma. We find that depletion of immunoglobulin M is associated with the most substantial loss of virus neutralization, followed by immunoglobulin G. This observation may help design efficient antibody-based COVID-19 therapies and may also explain the increased susceptibility to SARS-CoV-2 of autoimmune patients receiving therapies that impair the production of immunoglobulin M (IgM).
Research News: Understanding antibodies at play in long-lasting COVID-19 immunity
"Structure-function studies of the C3/C5 epimerases and C4 reductases of the Campylobacter jejuni capsular heptose modification pathways." - Creuzenet Lab
 Many bacteria produce polysaccharide-based capsules that protect them from environmental insults and play a role in virulence, host invasion, and other functions. Understanding how the polysaccharide components are synthesized could provide new means to combat bacterial infections. We have previously characterized two pairs of homologous enzymes involved in the biosynthesis of capsular sugar precursors GDP-6-deoxy-D-altro-heptose and GDP-6-OMe-L-gluco-heptose in Campylobacter jejuni. However, the substrate specificity and mechanism of action of these enzymes – C3 and/or C5 epimerases DdahB and MlghB and C4 reductases DdahC and MlghC – are unknown. Here, we demonstrate that these enzymes are highly specific for heptose substrates, using mannose substrates inefficiently with the exception of MlghB. We show that DdahB and MlghB feature a jellyroll fold typical of cupins, which possess a range of activities including epimerizations, GDP occupying a similar position as in cupins. DdahC and MlghC contain a Rossman fold, a catalytic triad and a small C-terminal domain typical of short-chain dehydratase reductase enzymes. Integrating structural information with site-directed mutagenesis allowed us to identify features unique to each enzyme and provide mechanistic insight. In the epimerases, mutagenesis of H67, D173, N121, Y134 and Y132 suggested the presence of alternative catalytic residues. We showed that the reductases could reduce GDP-4-keto-6-deoxy-mannulose without prior epimerization though DdahC preferred the pre-epimerized substrate, and identified T110 and H180 as important for substrate specificity and catalytic efficacy. This information can be exploited to identify inhibitors for therapeutic applications or to tailor these enzymes to synthesise novel sugars useful as glycobiology tools.
Many bacteria produce polysaccharide-based capsules that protect them from environmental insults and play a role in virulence, host invasion, and other functions. Understanding how the polysaccharide components are synthesized could provide new means to combat bacterial infections. We have previously characterized two pairs of homologous enzymes involved in the biosynthesis of capsular sugar precursors GDP-6-deoxy-D-altro-heptose and GDP-6-OMe-L-gluco-heptose in Campylobacter jejuni. However, the substrate specificity and mechanism of action of these enzymes – C3 and/or C5 epimerases DdahB and MlghB and C4 reductases DdahC and MlghC – are unknown. Here, we demonstrate that these enzymes are highly specific for heptose substrates, using mannose substrates inefficiently with the exception of MlghB. We show that DdahB and MlghB feature a jellyroll fold typical of cupins, which possess a range of activities including epimerizations, GDP occupying a similar position as in cupins. DdahC and MlghC contain a Rossman fold, a catalytic triad and a small C-terminal domain typical of short-chain dehydratase reductase enzymes. Integrating structural information with site-directed mutagenesis allowed us to identify features unique to each enzyme and provide mechanistic insight. In the epimerases, mutagenesis of H67, D173, N121, Y134 and Y132 suggested the presence of alternative catalytic residues. We showed that the reductases could reduce GDP-4-keto-6-deoxy-mannulose without prior epimerization though DdahC preferred the pre-epimerized substrate, and identified T110 and H180 as important for substrate specificity and catalytic efficacy. This information can be exploited to identify inhibitors for therapeutic applications or to tailor these enzymes to synthesise novel sugars useful as glycobiology tools.
"Innate Immune Amnesia" - Haeryfar Lab
 Measles virus (MeV) binds, infects and kills CD150+ memory T-cells, leading to "immune amnesia". Whether MeV targets innate, memory-like T-cells is unknown. We demonstrate that human peripheral blood and hepatic MAIT and iNKT cells express surprisingly high levels of CD150, more than any other lymphocyte subsets, including central and effector memory T-cells. Furthermore, exposing MAIT cells to MeV results in their efficient infection and rapid apoptosis. This constitutes the first report of direct MAIT cell infection by a viral pathogen. Given MAIT cells' antimicrobial properties, their elimination by MeV may contribute to measles-induced immunosuppression and heightened vulnerability to unrelated infections.
Measles virus (MeV) binds, infects and kills CD150+ memory T-cells, leading to "immune amnesia". Whether MeV targets innate, memory-like T-cells is unknown. We demonstrate that human peripheral blood and hepatic MAIT and iNKT cells express surprisingly high levels of CD150, more than any other lymphocyte subsets, including central and effector memory T-cells. Furthermore, exposing MAIT cells to MeV results in their efficient infection and rapid apoptosis. This constitutes the first report of direct MAIT cell infection by a viral pathogen. Given MAIT cells' antimicrobial properties, their elimination by MeV may contribute to measles-induced immunosuppression and heightened vulnerability to unrelated infections.
https://pubmed.ncbi.nlm.nih.gov/33310870/
https://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1009071








