Western Clinical Trials Leadership Program

Do you want to transform your trial concept into a fundable, execution-ready protocol?
The Western Clinical Trials Leadership Program is a new, competitive opportunity that brings together a select cohort of trialists ready to develop high-impact explanatory or pragmatic randomized trial concepts into well-designed, execution-ready protocols. Explore the differences between explanatory and pragmatic clinical trials.
With an expert team by your side, this 20-month program provides end-to-end guidance, expertise, infrastructure, mentorship, and structured learning from experienced trialists to support the transformation of your trial concept into an execution-ready protocol. The program starts in October 2026.

Applications Close April 30, 2026.
Start Your Application
Funding Support
Two matching streams are available within the program, each providing structured trial support services valued at up to $50,000.
Participants selected for the Open-Match stream will receive up to $50,000 in trial support services — $25,000 provided by Western, contingent upon an additional $25,000 in matching support from their department, faculty, or comparable source.
Participants selected for the AMOSO-Match stream will also receive up to $50,000 in trial support services, jointly supported by Western and AMOSO.
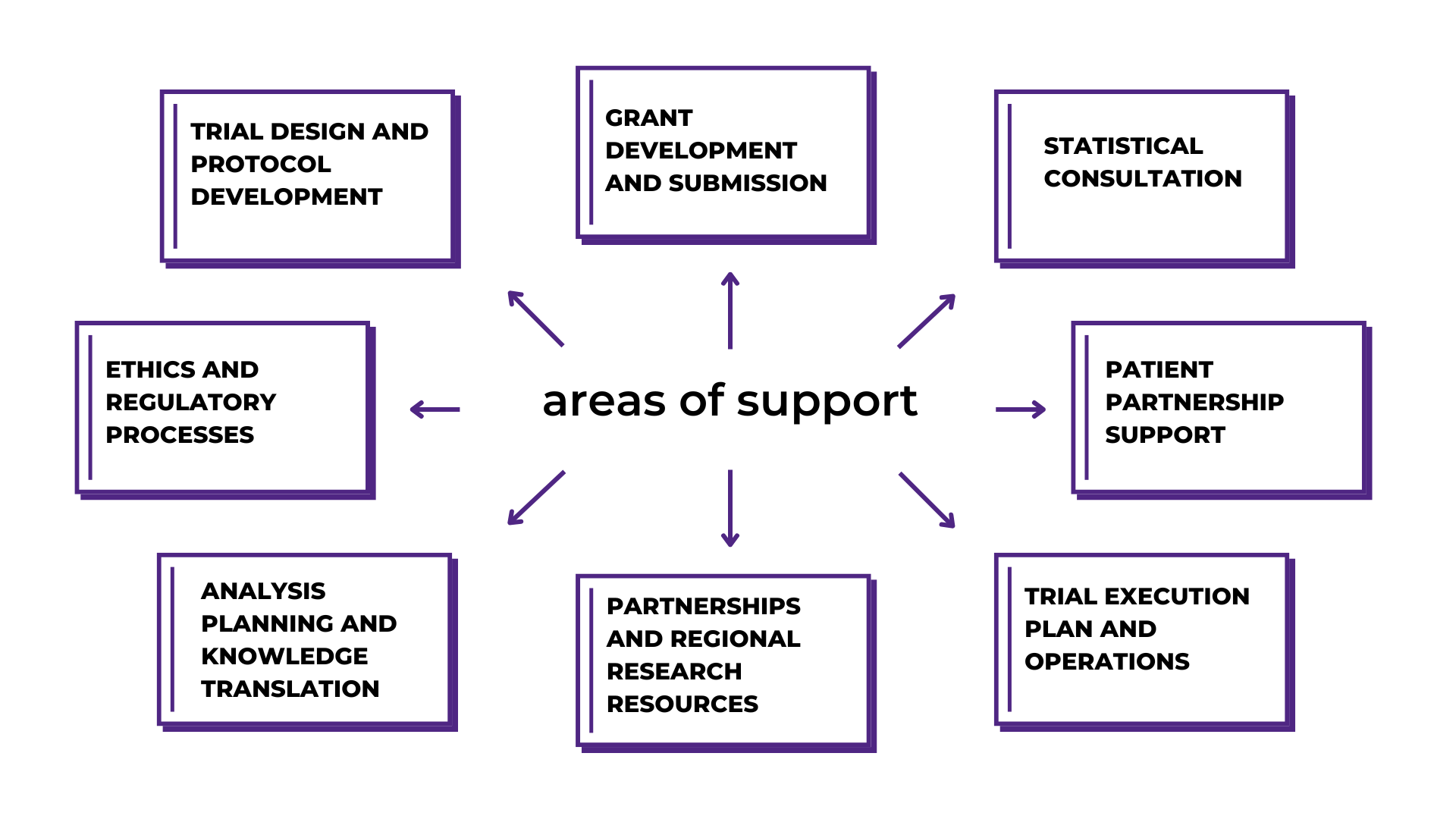
Areas of Support
With a dedicated expert team at your side, you can go further in your research and receive guidance in:
Are You Eligible?
We are seeking motivated applicants who hold primary appointments within any Faculty at Western University and can commit to a minimum of 7 hours per week over 20 months to advance their trial.
Open-Match Stream
- Propose an explanatory or pragmatic randomized clinical trial concept.
- Secure confirmation of matching support up to $25,000 from their Department, Faculty, or other sources.
AMOSO-Match Stream
- Propose a pragmatic randomized trial concept
- Member of AMOSO
Eligible Sources of Matching Funds
Matching contributions may include existing funds that investigators can allocate toward the development of their proposed clinical trial. Examples of eligible sources include:
- Departmental or Faculty support
- Investigator start-up funds
- Unrestricted research accounts
- Funds from service agreements with industry partners (provided they are unrestricted for research use). Please note that the use of industry-related funds may limit access to certain large publicly funded healthcare databases and may require a plan to manage potential conflicts of interest.
- External grants supporting preliminary or pilot work related to the proposed trial. Trials that are already funded are not eligible.
- Career awards, where permitted by sponsor guidelines (generally referring to the research or operating grant component of the award rather than the salary support portion)
Key Dates
-
✅Applications open March 1, 2026
-
🟥Application deadline April 30, 2026
-
👉Program starts October 2026








