Feature: 900,000+ COVID-19 tests processed by PaLM Microbiology Division
By Alexandra Burza, MMJC'19
Between the imminent border closures and news of stay-at-home orders, for many, March 18, 2020 was defined by overwhelming uncertainty. But at the Schulich School of Medicine & Dentistry’s Pathology and Laboratory Medicine (PaLM) clinical labs, decisive action was being taken to support the COVID-19 testing needs of Southwestern Ontario.
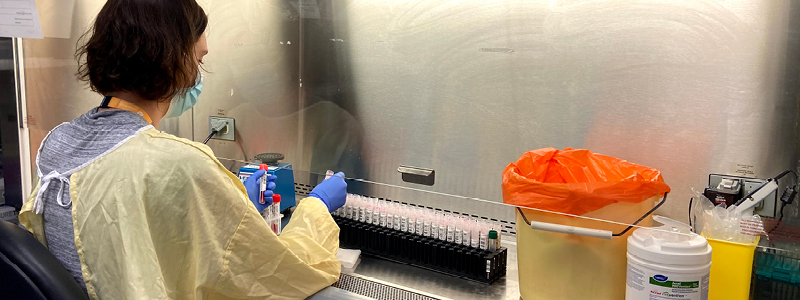
On that day, the PaLM Microbiology lab performed, processed and reported its first test for the novel coronavirus. During the course of the next 14 months, the lab processed more than 900,000 tests for seven public health units, including hospitals and long-term care facilities, from Owen Sound to Windsor.
While the lab already offered molecular diagnostic services before the pandemic, many adaptations and fast growth were necessary to implement and facilitate COVID-19 testing operations.
“We used to, in a particularly bad influenza season, test 30, maybe 50 samples a day. Now, when we have a really busy day, we’re processing 10,000 COVID-19 tests,” said Dr. Johan Delport, Division Head, Microbiology and Assistant Professor, Pathology and Laboratory Medicine.
In just one year of COVID-19 testing, PaLM processed the equivalent to over 150 years of regular seasonal influenza tests.
As demand for SARS-CoV-2 testing quickly grew worldwide, the lab was met with the now all-too-familiar issue of supply shortages.
“Everything we use for testing is made for one-time use: the swabs, plates, reagents, and we are competing for the same materials with every other lab,” explained Dr. Ana Cabrera, Molecular Microbiology Section Head and Assistant Professor, Pathology and Laboratory Medicine.
“There was definitely a point in time where the system hit a wall, because manufacturers and producers were already struggling to adapt to very fast change without preparation, while our demand for supplies was only getting bigger.”

Delport and Cabrera recalled a critical point in time last spring, when they had kits available for 100 tests a day, but only enough swabs for 80. The team overcame the shortage by sourcing swabs from dozens of unknown vendors to test against the swab they knew would be correct for this indication.
“We had this constant dynamic of looking for stuff, trying to source the various test components. It’s like cooking a meal; if one ingredient is not in stock, you can’t complete the meal,” Delport explained.
Whether it was slightly adjusting extraction protocols or the shape of a testing plate based on equipment availability, the lab was proactive in managing possible shortages and managed to test continuously without interruptions and impact to the other activities and diagnostic services.
“The biggest challenge was that there was no time to settle, to perfect our logistics,” Cabrera said. “This is happening now, and we had to have a system in place right away to provide this testing support. We needed to be able to adapt quickly, and our teams rose to the challenge.”
While operations never ceased, capacity for testing at the Lab was still limited for the first few months of the pandemic. As a leader in infection control protocols, Delport recalls having to make difficult decisions as to who could receive a COVID-19 test.
“In the beginning, it was incredibly emotional. And if I think about it, I still get tears in my eyes, because I had to say ‘no’ to patients and their families who needed tests. But the primary focus was always to keep hospitals and ICUs open.”
As they began to overcome challenges related to supply procurement, PaLM turned their attention to quickly growing their testing capacity. The technical team, responsible for processing and analyzing the tests, grew to more than double what it had been before the pandemic. Within three months, a new lab space was constructed to accommodate the additional staff and instrumentation.
Another important logistical decision which helped improve testing capacity was the use of specimen pooling, which involves testing multiple samples together. Only in the case of a positive result do all the pooled samples need to be re-tested to identify the COVID-positive case.
“We were one of the first labs in Ontario to go live with specimen pooling. There’s a lot of reasons why we decided to go with pooling; of course, primarily the constraints on consumables and instrumentation,” Cabrera explained “There is some risk, but also a lot of benefits if you can find the ideal balance on how many samples to pool together.”
Now, with increased positivity rates across many communities, Cabrera says this method is less likely to be utilized, reserved for populations that have a lesser likelihood of infection such as those who require COVID-19 screening for work but do not exhibit symptoms.
With cases currently at an all-time high in Ontario, the teams at the lab are working as hard as ever to keep up with testing demand. Delport says that the commitment of technicians and microbiologists is tremendous, with many working long hours in the early morning and late evening to get the job done.
“It’s important to highlight how dedicated this group is to providing an excellent service to Southwestern Ontario,” he said. “We are so used to seeing the negative news lately, but their dedication is a major positive. They’ve been here, ready to work, every single day, and we really are a big working family that cares for each other.”








