The London Regional Proteomics Centre (LRPC) comprises six core facilities operated through the Department of Biochemistry, in the Schulich School of Medicine & Dentistry at Western University.
These constituent core facilities provide service and instrumentation and will work cooperatively to fulfill your research needs in the area of proteomics, protein structure and protein function.
Functional Proteomics Facility (FPF)
Medical Sciences Building, Room 323
Email: BioCORE@uwo.ca
The FPF is the most common entry point for researchers requiring analysis of complex mixtures of proteins, i.e. proteomics. Equipment is available for the fractionation of these complex biological mixtures using chromatographic and electrophoretic procedures and for the preparation of protein spots or fractions for identification/analysis. Normally, samples will then be passed on to the appropriate mass spectrometry core facility for that purpose.
Instrumentation housed within this facility includes a Beckman Biomek liquid handler, a BioTek Synergy plate reader, PE VICTOR 1420-40 multilabel counter, an infrared imaging system, a benchtop ultracentrifuge, an FPLC system, instrumentation for 2D electrophoresis, ProExpress Imaging System with Progenesis 2D analysis software, a spot-picker for excision of protein spots from gels, MASSPrep for automated proteolytic digestion of proteins and an LC-MALDIPrep that includes a capillary HPLC integrated with a MALDI spotter.
Biological mass Spectrometry Laboratory (BMSL)
Siebens Drake Research Institute, Room G31A
Email: BioCORE@uwo.ca
 The BMSL features mass spectrometers of several types. These instruments allow the detailed analysis of protein mass and peptide composition through various approaches. Some of the works performed at BMSL include: mass determination of polar and/or high molecular weight molecules including peptides, proteins, glycyoproteins, etc.; sequence determination of peptides and peptides from proteins digests; protein identification in a mixture; and types and sites of protein modifications.
The BMSL features mass spectrometers of several types. These instruments allow the detailed analysis of protein mass and peptide composition through various approaches. Some of the works performed at BMSL include: mass determination of polar and/or high molecular weight molecules including peptides, proteins, glycyoproteins, etc.; sequence determination of peptides and peptides from proteins digests; protein identification in a mixture; and types and sites of protein modifications.
Biomolecular Nuclear Magnetic Resonance Facility (BIONMR)
Medical Sciences Building, Room 309
Email: BioCORE@uwo.ca
 The Biomolecular NMR Facility houses state-of-the-art nuclear magnetic resonance (NMR) instrumentation and provides expertise required for the characterization of proteins, nucleic acids, carbohydrates and small molecules at the atomic level. A large number of biological NMR experiments and analyses software are available allowing the three-dimensional structure of proteins to be determined in solution, detailed characterization of protein-protein and protein-ligand interactions, and the identification of conformational changes, protein motions (dynamics) and folding processes.
The Biomolecular NMR Facility houses state-of-the-art nuclear magnetic resonance (NMR) instrumentation and provides expertise required for the characterization of proteins, nucleic acids, carbohydrates and small molecules at the atomic level. A large number of biological NMR experiments and analyses software are available allowing the three-dimensional structure of proteins to be determined in solution, detailed characterization of protein-protein and protein-ligand interactions, and the identification of conformational changes, protein motions (dynamics) and folding processes.
The facility offers training and technical expertise for researchers familiar with NMR techniques, those requiring assistance and first-time users. Researchers may also submit samples for expert data acquisition, analysis and interpretation.
MALDI Mass Spectrometry Facility (MALDI)
Seibens Drake Research Institute, Room G31
BioCORE@uwo.ca

Biomolecular Interactions and Conformations Facility (BICF)
Medical Sciences Building, Room 323
Email: BioCORE@uwo.ca
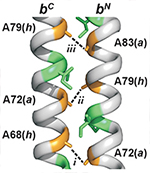 .
.
Macromolecular Crystallography Facility (MCF)
Medical Sciences Building, Room 333
Email: BioCORE@uwo.ca
 The MCF provides equipment, supplies, and expertise essential for determination of the three-dimensional structures of purified proteins through X-ray crystallography. Equipment includes a crystallization robot, an X-ray generator, and an area detector. Advice and technical assistance in sample preparation and experimentation is provided as needed for clients who wish to conduct the work themselves.
The MCF provides equipment, supplies, and expertise essential for determination of the three-dimensional structures of purified proteins through X-ray crystallography. Equipment includes a crystallization robot, an X-ray generator, and an area detector. Advice and technical assistance in sample preparation and experimentation is provided as needed for clients who wish to conduct the work themselves.
Alternatively, the MCF provides a full crystallization service; clients may submit samples for crystallization trials, and crystallization condition refinement, carried out by the Facility Manager.
COVID-19 Information for users of the LRPC
How We Can Help
LRPC facilities are accessible to users from Western University, external academic communities, and industrial settings. Users can perform their own sample analyses or use the services of qualified facility staff.

For further information on how the core facilities of the LRPC can accommodate your research goals, please contact us by email at: BioCORE@uwo.ca